Purpose
The purpose of the Public Review is to develop widespread consensus for the proposed standard by allowing for broad comment by the general public.
Scope
The deliverables are the standards document posting package, including electronic metadata as appropriate.
Prerequisites
The draft standard should include the following elements:
- Concept maps for key clinical concepts
- Metadata displays
- SDTM, CDASH and ADaM models or examples, as appropriate
Definitions
List terms (abbreviations, acronyms, technical jargon) referenced in this Work Instruction useful at adding clarity. Put "Not applicable." if this section is not applicable.
| Term | Definition |
|---|---|
| FDA | Food and Drug Administration The FDA is responsible for protecting the public health by ensuring the safety, efficacy, and security of human and veterinary drugs, biological products, |
| GGG | Global Governance Group Responsible for reviewing and approving all draft standards. This group is composed of representatives from each of the foundational teams who have been empowered to review and approve modeling decisions on behalf of their team. The GGG meetings are open to any CDISC team member. |
Roles & Responsibilities
Lists all roles and their responsibilities relevant to this Work Instruction.
| Role | Responsibilities |
|---|---|
| Project Manager |
|
| Technical writer |
|
| GGG |
|
| SRC |
|
| FDA |
|
Instructions
Describe each step to be taken to complete the scope of this Work Instruction in the prescribed sequence. Use active verb in the Instructions column.
| Step # | Role | Instructions |
|---|---|---|
| 1.0 | Technical Writer | Posts draft standard package for public review |
| 2.0 | Project Manager | Responds to Public Review comments and update the draft document |
| 3.0 | FDA | Reviews the standard |
| 4.0 | Project Manager | Responds to FDA comments and update the draft document |
| 5.0 | Project Manager | Publishes material ready for GGG |
| 6.0 | GGG | Meets for publication approval |
| 7.0 | GGG | Signs off for publication approval |
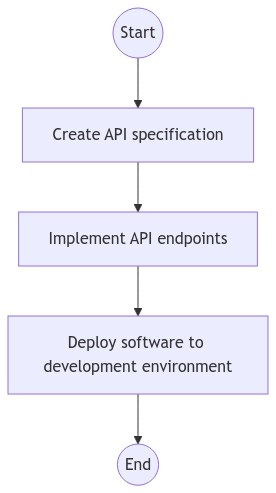
General Workflow
Include a general workflow diagram that summarizes this Work Instruction.
References
List references pertinent to this Work Instruction, e.g., COP-001.