- Created by Lou Ann Kramer, last modified on Jan 11, 2023
You are viewing an old version of this page. View the current version.
Compare with Current View Page History
« Previous Version 54 Next »
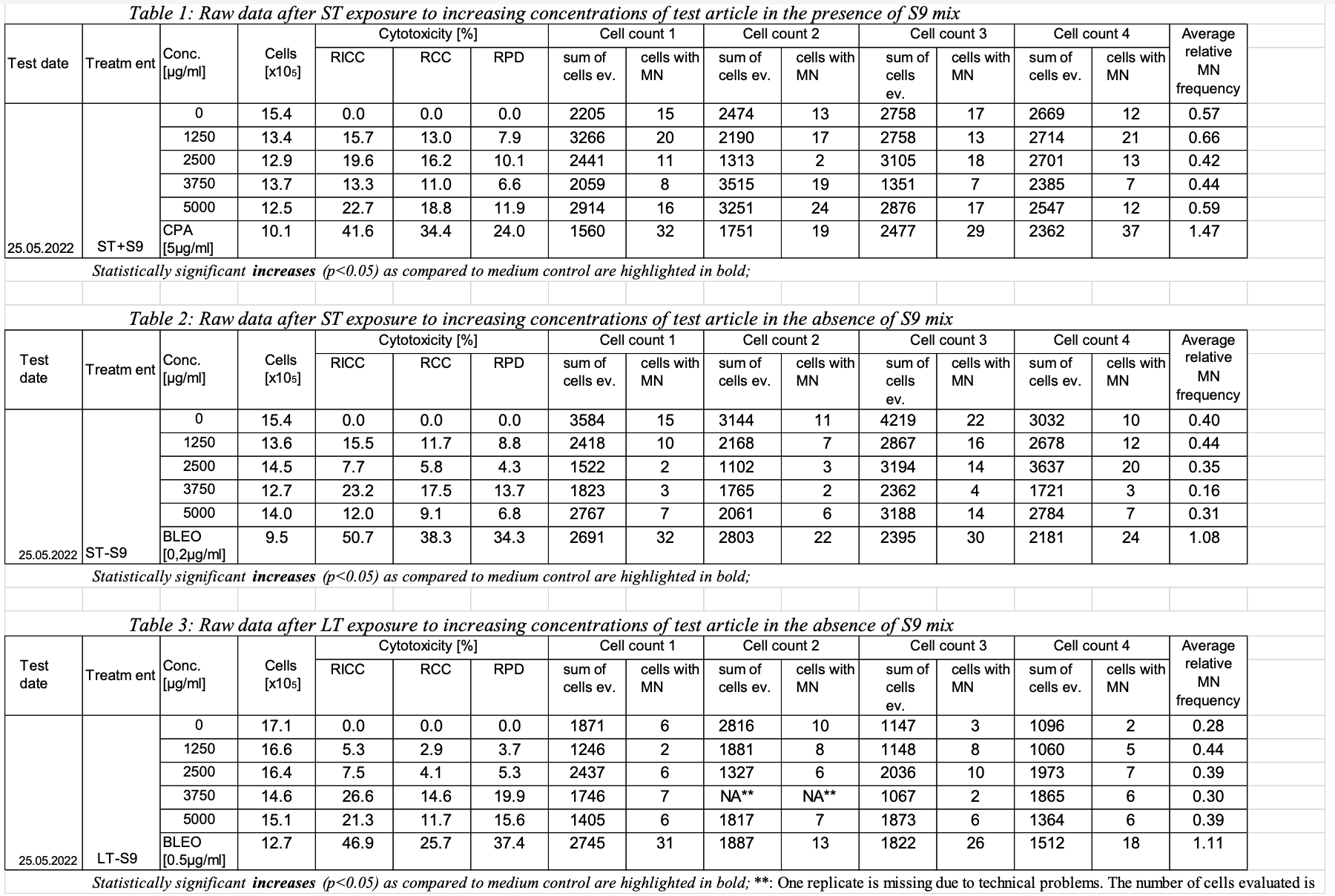
This is an example showing trial design and results data of Study #123 for the determination of the in vitro genotoxicity potential of 10 tobacco products in the in vitro Micronucleus Assay
- red font - indicates potential for CT code lists
- green font - links to other domains
- purple font - to be discussed
- Assumption: The intent of this dataset is to provide a summary of trial (study) information. This is not subject-level data.
- Assumption: A Trial (study) can have more than one assay type
- Assumption: ASSAYID value of ALL indicates that it applies to all assays in the study
- Assumption: SPTOBID (product identifier) should be added at the trial set level (tx.xpt)
- This allows for studies where there are multiple products and different product(s) per trial set, one record for each product that is being tested in each particular trial set (tx.xpt)
Row | STUDYID | ASSAYID | DOMAIN | TSSEQ | TSGRPID | TSPARMCD | TSPARM | TSVAL | TSVALNF |
|---|---|---|---|---|---|---|---|---|---|
| 1 | 123 | MNvit | TS | 1 | GLPTYP | Good Laboratory Practice Type | FDA | ||
| 2 | 123 | MNvit | TS | 2 | GLPTYP | Good Laboratory Practice Type | OECD | ||
| 3 | 123 | MNvit | TS | 1 | STSTDTC | Study Start Date | 2022-05-25 | ||
| 4 | 123 | MNvit | TS | 1 | STITLE | Study Title | Determination of the in vitro genotoxicity potential of 10 tobacco products in the in vitro Micronucleus Assay | ||
| 5 | 123 | MNvit | TS | 1 | SNDIGVER | SEND Implementation Guide Version | TOBACCO IMPLEMENTATION GUIDE VERSION 1.0 | ||
| 6 | 123 | MNvit | TS | 1 | SNDCTVER | SEND Controlled Terminology Version | SEND Terminology 2021-09-30 | ||
| 7 | 123 | MNvit | TS | 1 | SSPONSOR | Sponsor Organization | Example Sponsor Inc. | ||
| 8 | 123 | MNvit | TS | 1 | SPREFID | Sponsor's Study Reference ID | NOT APPLICABLE | ||
| 9 | 123 | MNvit | TS | 1 | 1 | TSTFNAM | Test Facility Name | Example Tox Lab Name | |
| 10 | 123 | MNvit | TS | 1 | 1 | TSTFLOC | Test Facility Location | 10 Somewhere Street, Montgomery, AL 10000 | |
| 11 | 123 | MNvit | TS | 1 | 1 | TFCNTRY | Test Facility Country | USA | |
| 12 | 123 | MNvit | TS | 1 | 1 | STDIR | Study Director | Dr. R. Smith | |
| 13 | 123 | MNvit | TS | 1 | GLPFL | GLP Flag | Y | ||
| 14 | 123 | MNvit | TS | 1 | ASTD | Assay Standard | OECD Test No. 487 | ||
| 15 | 123 | MNvit | TS | 1 | ASTDV | Assay Standard Version | 2016-07-29 | ||
| 16 | 123 | MNvit | TS | 1 | SSTYP | Study Type | GENOTOXICITY IN VITRO | ||
| 17 | 123 | MNvit | TS | 1 | SSSTYP | Study Sub Type | In Vitro Micronucleus | ||
| 18 | 123 | MNvit | TS | 1 | SPECIES | Species | Homo Sapiens | ||
| 19 | 123 | MNvit | TS | 1 | ?? | Test System? | TK6 Lymphoblastoid Suspension Cells | ||
| 20 | 123 | MNvit | TS | 1 | SPTOBID | Sponsor defined tobacco product identifier | CIG01a | ||
| 21 | 123 | MNvit | TS | 1 | 1? | TCVAR1 | Product testing conditions | PTTSTCND | |
| 22 | 123 | MNvit | TS | 1 | 1? | TCVRVAL1 | E-Liquid Standard (Long Term) Condition |
- During CT definition/reviews will decide appropriate TXPARM and TXVAL; Treatment duration may be controlled; For now, we just include good example values based on our experience
- Assumption: The Trial Sets (TX) domain provides the list of distinct sets of subjects having different experimental factors, treatment factors, inherent characteristics, or distinct sponsor designations as specified in the trial design.
- Where is TK6 cell type? is this test system (see below)
- needs to be allowed to vary down to the well level / result level
A1: 
A2: 
| Row | STUDYID | ASSAYID | DOMAIN | SETCD | SET (what sponsor calls it) | TXSEQ | TXPARMCD | TXPARM | TXVAL |
|---|---|---|---|---|---|---|---|---|---|
| 123 | MNvit | TX | A1 (table 1, row 1, ST exposure with S9) | METACT | Metabolic Activation (should there be two parms? Presence, type)? | +S9 | |||
| METACTFL | Y/N presence of metabolic activation | ||||||||
| 123 | MNvit | TX | A1 | TRTDRTRG | Treatment Duration target. (how do we show 3-6 hour range? start/end, target and tolerance?, one text field not-analyzable) | 3 | |||
| 123 | MNvit | TX | A1 | TRTDRTOL | Treatment Duration Tolerance | ||||
| 123 | MNvit | TX | A1 | TRTDURU | Treatment Duration Unit (this is for both TRTDURT, TRTDURTOL) | H | |||
| 123 | MNvit | TX | A1 | INTRVN | name of the intervention article (Tobacco ProdA, Bleomycin or Cyclophosphamid A) | Tobacco ProdA | |||
| 123 | MNvit | TX | A1 | ITVTYPE | type of intervention article choices of values: product; negative control; positive control | Product | |||
| 123 | MNvit | TX | A1 | ITVCONC | Concentration of intervention article | 0 | |||
| 123 | MNvit | TX | A1 | ITVCONCU | Concentration Unit | ug/ml | |||
| 123 | MNvit | TX | A2 (table 1, row 2) | METACT | Metabolic Activation (should there be two parms? Presence, type)? | +S9 | |||
| 123 | MNvit | TX | A2 | TRTDRTRG | Treatment Duration target. (how do we show 3-6 hour range? start/end, target and tolerance?, one text field not-analyzable) | 3 | |||
| 123 | MNvit | TX | A2 | TRTDRTOL | Treatment Duration Tolerance | ||||
| 123 | MNvit | TX | A2 | TRTDURU | Treatment Duration Unit (this is for both TRTDURT, TRTDURTOL) | H | |||
| 123 | MNvit | TX | A2 | INTRVN | name of the intervention article | Tobacco ProdA | |||
| 123 | MNvit | TX | A2 | ITVTYPE | type of intervention article | Product | |||
| 123 | MNvit | TX | A2 | ITVCONC | Concentration of i a | 1250 | |||
| 123 | MNvit | TX | A2 | ITVCONCU | Concentration Unit | ug/ml | |||
| ... | |||||||||
| Rows 1-4: | Show the records for the product identifiers for the tobacco product identified in SPTOBID. These records are categorized as product identifiers by TOCAT = PRODUCT IDENTIFIERS. |
|---|---|
| Rows 5-10: | Show the records for the product descriptors for the tobacco product identified in SPTOBID. These records are categorized as product identifiers by TOCAT = PRODUCT DESCRIPTOR. |
to.xpt
Row | STUDYID | DOMAIN | SPTOBID | TOSEQ | TOTESTCD | TOTEST | TOCAT | TOORRES | TOORRESU | TOSTRESC | TOSTRESN | TOSTRESU |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | TOB07 | TO | CIG01a | 1 | TBPRDCAT | Tobacco Product Category | PRODUCT IDENTIFIER | Cigarette | Cigarette | |||
| 2 | TOB07 | TO | CIG01a | 2 | TBPRSCAT | Tobacco Product Subcategory | PRODUCT IDENTIFIER | Filtered, Combusted | Filtered, Combusted | |||
| 3 | TOB07 | TO | CIG01a | 3 | MANUF | Manufacturer | PRODUCT IDENTIFIER | Joes Cigs USA | Joes Cigs USA | |||
| 4 | TOB07 | TO | CIG01a | 4 | TRADENAM | Trade Name | PRODUCT IDENTIFIER | Treetop Menthol King Size | Treetop Menthol King Size | |||
| 5 | TOB07 | TO | CIG01a | 5 | PACKTYP | Package Type | PRODUCT DESCRIPTOR | HARD PACK | HARD PACK | |||
| 6 | TOB07 | TO | CIG01a | 6 | PRDQUAN | Product Quantity | PRODUCT DESCRIPTOR | 20 | CIGARETTE | 20 | 20 | CIGARETTE |
| 7 | TOB07 | TO | CIG01a | 7 | LENGTH | Length | PRODUCT DESCRIPTOR | 86.0 | mm | 86.0 | 86.0 | mm |
| 8 | TOB07 | TO | CIG01a | 8 | CIRCUMF | Circumference | PRODUCT DESCRIPTOR | 26.0 | mm | 26.0 | 26.0 | mm |
| 9 | TOB07 | TO | CIG01a | 9 | VENTLTN | Ventilation | PRODUCT DESCRIPTOR | 10.0 | % | 10.0 | 10.0 | % |
| 10 | TOB07 | TO | CIG01a | 10 | CHARFLAV | Characterizing Flavor | PRODUCT DESCRIPTOR | MENTHOL | MENTHOL |
The Test Conditions (TC) dataset allows for listing the attributes of the testing regimens used in tobacco product testing when the details are too long and variable for representing as a single value in the PTTSTCND variable in a PT dataset.
The example below shows the attributes of the testing regimen identified as PTTSTCND="E-Liquid Standard Aerosol Condition" (see e-liquid stability study pt.xpt example). Note that the combination of TCVAR1="PTTSTCND" and TCVRVAL1="E-Liquid Standard (Long Term) Condition" identifies the specific testing regimen shown in PTTSTCND as the focus of the testing regimen parameters listed in TCPARMCD/TCPARM. TCVAL and TCVALU show the specific values and units (respectively, where applicable) for the given parameter.one testing regimen shown in the PT stability study above.
(We've added TCVALU which was not originally part of the TC draft domain. May propose elevating this structure to a "Trial Lookup" observational class, within which some domains would allow for TCVALU)
tc.xpt
tc.xpt
| Row | STUDYID | DOMAIN | RDOMAIN | TCVAR1 | TCVRVAL1 | TCSEQ | TCGRPID | TCPARMCD | TCPARM | TCVAL | TCVALU |
|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | TOB07 | TC | PT | PTTSTCND | E-Liquid Standard (Long Term) Condition | 1 | PUFFPROF | Puff Profile | SQUARE | ||
| 2 | TOB07 | TC | PT | PTTSTCND | E-Liquid Standard (Long Term) Condition | 2 | PUFFDUR | Puff Duration | 1.25 | sec | |
| 3 | TOB07 | TC | PT | PTTSTCND | E-Liquid Standard (Long Term) Condition | 3 | PUFFINT | Puff Interval | 3 | PUFF/min | |
| 4 | TOB07 | TC | PT | PTTSTCND | E-Liquid Standard (Long Term) Condition | 4 | PUFFBLCK | Puff Block | 25 | % | |
| 5 | TOB07 | TC | PT | PTTSTCND | E-Liquid Standard (Long Term) Condition | 5 | NUMPUFF | Total Number of Puffs | 200 | PUFF | |
| 6 | TOB07 | TC | PT | PTTSTCND | E-Liquid Standard (Long Term) Condition | 6 | PUFFVOL | Puff Volume | 10 | mL | |
| 7 | TOB07 | TC | PT | PTTSTCND | E-Liquid Standard (Long Term) Condition | 7 | PUFFRNG | Puff Range | 100-200 | ||
| 8 | TOB07 | TC | PT | PTTSTCND | E-Liquid Standard (Long Term) Condition | 8 | 1 | PUFFPAUS | Puff Pause | 60 | s |
| 9 | TOB07 | TC | PT | PTTSTCND | E-Liquid Standard (Long Term) Condition | 9 | 1 | PUFFPINT | Puff Pause Interval | 10 | PUFF |
A1: 
A2: 
| Row | STUDYID | ASSAYID | DOMAIN | TXSETCD | GTSEQ | GTTESTCD | GTTEST | GTCELLEV (cells evaluated) | GTORRES | GTORRESU | GTSTRESC | GTSTRESN | GTSTRESU | GTDTC |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 123 | MNvit | GT | A1 | 1 | RICC | Relative Increase in Cell Count | 0 | 0 | % | 0 | 0 | % | 2022-05-25 |
| 2 | 123 | MNvit | GT | A1 | 2 | RCC | Relative Cell Count | 154 | 0 | % | 0 | 0 | % | 2022-05-25 |
| 3 | 123 | MNvit | GT | A1 | 3 | RPD | Relative Population Doubling | 154 | 0 | % | 0 | 0 | % | 2022-05-25 |
| 4 | 123 | MNvit | GT | A1 | 4 | MNCELLS | Micronucleated Cells | 2205 | 15 | Cells | 15 | 15 | Cells | 2022-05-25 |
| 5 | 123 | MNvit | GT | A1 | 5 | MNCELLS | Micronucleated Cells | 2474 | 13 | Cells | 13 | 13 | Cells | 2022-05-25 |
| 6 | 123 | MNvit | GT | A1 | 6 | MNCELLS | Micronucleated Cells | 2758 | 17 | Cells | 17 | 17 | Cells | 2022-05-25 |
| 7 | 123 | MNvit | GT | A1 | 7 | MNCELLS | Micronucleated Cells | 2669 | 12 | Cells | 12 | 12 | Cells | 2022-05-25 |
| 8 | 123 | MNvit | GT | A1 | 8 | AVGREL | Average Relative MN Frequency | 0.57 | % | 0.57 | 0.57 | % | 2022-05-25 | |
| 9 | 123 | MNvit | GT | A2 | 1 | RICC | Relative Increase in Cell Count | 134 | 15.7 | % | 15.7 | 15.7 | % | 2022-05-25 |
| 10 | 123 | MNvit | GT | A2 | 2 | RCC | Relative Cell Count | 134 | 13.0 | % | 13.0 | 13.0 | % | 2022-05-25 |
| 11 | 123 | MNvit | GT | A2 | 3 | RPD | Relative Population Doubling | 134 | 7.9 | % | 7.9 | 7.9 | % | 2022-05-25 |
| 12 | 123 | MNvit | GT | A2 | 4 | MNCELLS | Micronucleated Cells | 3266 | 20 | Cells | 20 | 20 | Cells | 2022-05-25 |
| 13 | 123 | MNvit | GT | A2 | 5 | MNCELLS | Micronucleated Cells | 2190 | 17 | Cells | 17 | 17 | Cells | 2022-05-25 |
| 14 | 123 | MNvit | GT | A2 | 6 | MNCELLS | Micronucleated Cells | 2758 | 13 | Cells | 13 | 13 | Cells | 2022-05-25 |
| 15 | 123 | MNvit | GT | A2 | 7 | MNCELLS | Micronucleated Cells | 2714 | 21 | Cells | 21 | 21 | Cells | 2022-05-25 |
| 16 | 123 | MNvit | GT | A2 | 8 | AVGREL | Average Relative MN Frequency | 0.66 | % | 0.66 | 0.66 | % | 2022-05-25 | |
| ... |
- No labels