Page History
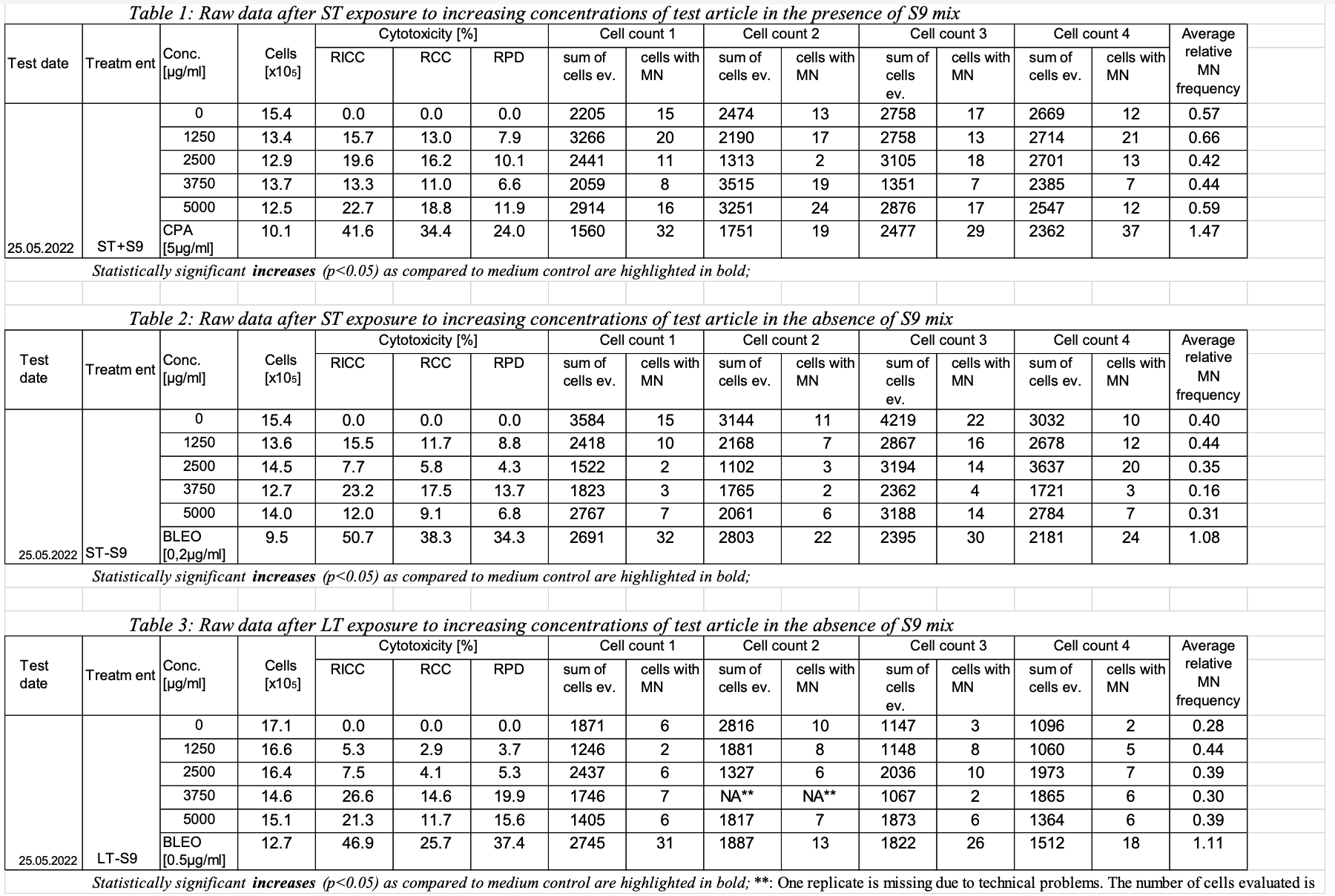
This is an example showing example shows a sample report table, trial design, and results data of Study #123 dataset for study 123 for the determination of the in vitro genotoxicity potential of 10 tobacco products in using the in vitro Micronucleus Assaymicronucleus assay.
| Expand | ||||||||
|---|---|---|---|---|---|---|---|---|
| Fold increase of MN over background | Significant increase Y/N | 1250 | 0.9 | 0.9973 | N | ||
2500 | 0.9 | 0.9517 | N | |||||
3750 | 0.6 | 0.2654 | N | |||||
5000 | 0.9 | 0.9895 | N | |||||
CPA [5µg/ml] | 3.1 | 0.0003 | Y | 1250 | 1.6 | 0.6408 | N | |
2500 | 1.3 | 0.8995 | N | |||||
3750 | 2.9 | 0.0106 | N | |||||
5000 | 1.0 | > 0.9999 | N | |||||
Bleo [0.2µg/ml] | 3.8 | 0.0002 | Y | 1250 | 0.7 | 0.5958 | N | |
2500 | 1.3 | 0.6378 | N | |||||
3750 | 0.5 | 0.1146 | N | |||||
5000 | 1.1 | 0.9929 | N | |||||
Bleo [0.5µg/ml] | 2.9 | 0.0047 | Y |
| Expand | ||
|---|---|---|
| ||
| |
| Dataset wrap | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
This example Trial Sets dataset shows information about the test conditions for set A1 and A2 in this example study. Sets A1 and A2 can be seen in the first and second rows respectively of the sample report Table 1 (above). For brevity, the TX dataset and the findings (GT) dataset do not show information for any other sets. Fully formed datasets for this example study would include information about the test conditions and findings for all sets.
| Dataset wrap | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
| Dataset wrap | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
| Dataset wrap | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Expand | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Row | STUDYID | ASSAYID | DOMAIN | TSSEQ | TSGRPID | TSPARMCD | TSPARM | TSVAL | TSVALNF | 1 | 123 | MNvit | TS | 1 | GLPTYP | Good Laboratory Practice Type | FDA | 2 | 123 | MNvit | TS | 2 | GLPTYP | Good Laboratory Practice Type | OECD | 3 | 123 | MNvit | TS | 1 | STSTDTC | Study Start Date | 2022-05-25 | 4 | 123 | MNvit | TS | 1 | STITLE | Study Title | Determination of the in vitro genotoxicity potential of 10 tobacco products in the in vitro Micronucleus Assay | 5 | 123 | MNvit | TS | 1 | SNDIGVER | SEND Implementation Guide Version | TOBACCO IMPLEMENTATION GUIDE VERSION 1.0 | 6 | 123 | MNvit | TS | 1 | SNDCTVER | SEND Controlled Terminology Version | SEND Terminology 2021-09-30 | 7 | 123 | MNvit | TS | 1 | SSPONSOR | Sponsor Organization | Example Sponsor Inc. | 8 | 123 | MNvit | TS | 1 | SPREFID | Sponsor's Study Reference ID | NOT APPLICABLE | 9 | 123 | MNvit | TS | 1 | 1 | TSTFNAM | Test Facility Name | Example Tox Lab Name | 10 | 123 | MNvit | TS | 1 | 1 | TSTFLOC | Test Facility Location | 10 Somewhere Street, Montgomery, AL 10000 | 11 | 123 | MNvit | TS | 1 | 1 | TFCNTRY | Test Facility Country | USA | 12 | 123 | MNvit | TS | 1 | 1 | STDIR | Study Director | Dr. R. Smith | 13 | 123 | MNvit | TS | 1 | GLPFL | GLP Flag | Y | 14 | 123 | MNvit | TS | 1 | ASTD | Assay Standard | OECD Test No. 487 | 15 | 123 | MNvit | TS | 1 | ASTDV | Assay Standard Version | 2016-07-29 | 16 | 123 | MNvit | TS | 1 | SSTYP | Study Type | GENOTOXICITY IN VITRO | 17 | 123 | MNvit | TS | 1 | SSSTYP | Study Sub Type | In Vitro Micronucleus | 18 | 123 | MNvit | TS | 1 | SPECIES | Species | Homo Sapiens |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Expand | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
| Rows 1-4: | Show the records for the product identifiers for the tobacco product identified in SPTOBID. These records are categorized as product identifiers by TOCAT = PRODUCT IDENTIFIERS. |
|---|---|
| Rows 5-10: | Show the records for the product descriptors for the tobacco product identified in SPTOBID. These records are categorized as product identifiers by TOCAT = PRODUCT DESCRIPTOR. |
to.xpt
Row
STUDYID
DOMAIN
SPTOBID
TOSEQ
TOTESTCD
TOTEST
TOCAT
TOORRES
TOORRESU
TOSTRESC
TOSTRESN
TOSTRESU
1
| title | tx.xpt (trial sets) |
|---|
- During CT definition/reviews will decide appropriate TXPARM and TXVAL; Treatment duration may be controlled; For now, we just include good example values based on our experience
- Assumption: The Trial Sets (TX) domain provides the list of distinct sets of subjects having different experimental factors, treatment factors, inherent characteristics, or distinct sponsor designations as specified in the trial design.
- Where is TK6 cell type? is this test system (see below)
- needs to be allowed to vary down to the well level / result level
- Assumption: SPDEVID (sponsor defined device identifier), and Test System should be added at the trial set level (tx.xpt). While it is possible to show a value in TS when there is only one device for a study, we will make it a rule that it goes in TX for consistency and cross-study analysis.
- This allows for studies where there are multiple products and different product(s) per trial set, one record for each product that is being tested in each particular trial set (tx.xpt)
- Assumption: SPDEVID and DUREFID should only be in TX (with the same value for all sets if there is only one device used)
- Assumption: SPDEVID (sponsor defined device identifier) should be added at the trial set level (tx.xpt). While it is possible to show a value in TS when there is only one device for a study, we will make it a rule that it goes in TX for consistency and cross-study analysis.
- This allows for studies where there are multiple products and different product(s) per trial set, one record for each product that is being tested in each particular trial set (tx.xpt)
- Do we need both SPDEVID AND DUREFID?
- What are good values (realistic) for "SET"?
- Check Essential Data list
A1:
A2:
A1
(table 1, row 1, ST exposure with S9)
A1
A1
A1
A1
A1
A1
Sponsor defined tobacco identifier
Exposure Type (See TIG NC workstream minutes 30-Jan here: Nonclinical)
Sample Type
(e.g. TPM, GVP, whole aerosol, whole smoke conditioned media, aqueous extracts, etc., see notes here: Nonclinical)
Name of the Intervention Article
can be: Tobacco ProdA, Bleomycin (positive control) or Cyclophosphamid A (positive control)
type of intervention article
choices of values: product; negative control; positive control
A2
(table 1, row 2, ST exposure with S9 at concentration 1250)
A2
Sponsor defined tobacco identifier
Exposure Type (See TIG NC workstream minutes 30-Jan here: Nonclinical)
Sample Type
(e.g. TPM, GVP, whole aerosol, whole smoke conditioned media, aqueous extracts, etc., see notes here: Nonclinical)
Name of the Intervention Article
can be: Tobacco ProdA, Bleomycin (positive control) or Cyclophosphamid A (positive control)
type of intervention article
choices of values: product; negative control; positive control
| title | du.xpt smoke regimen definition (a findings domain for device in-use properties) |
|---|
We use DU for smoking regimen. Note that a separate DI dataset will be needed to show identifying parameters of the "PUFFMASTER3K" smoking machine
- Details of the smoking regimen are represented as device in-use properties, linked to the stability data in PT above by matching values of PTREFID/DUREFID = "Medium Intensity Regimen" (We will update with a realistic value for the regimen, with input).
- Smoking regimen is represented in --REFID (we made up a value of "Medium Intensity Regimen"; we can update with something realistic)
- The smoking regimen is carried out by the smoking machine/device shown in SPDEVID, Sponsor defined device identifier, "PUFFMASTER3K"
du.xpt
Row
STUDYID
DOMAIN
SPDEVID
DUSEQ
DUREFID
DUGRPID
DUTESTCD
DUTEST
DUORRES
DUORRESU
DUSTRESC
DUSTRESN
DUSTRESU
PUFFDUR
1.25
sec
1.25
sec
PUFFINT
3
PUFF/min
3
PUFF/min
PUFFBLCK
25
%
25
%
200
PUFF
200
PUFF
10
mL
10
mL
100-200
100-200
60
s
60
s
10
PUFF
10
PUFF
Canadian Intense Regime
PUFFDUR
2.00
sec
2.00
sec
PUFFINT
4
PUFF/min
4
PUFF/min
Canadian Intense Regime
PUFFBLCK
0
%
0
%
200
PUFF
200
PUFF
10
mL
10
mL
Canadian Intense Regime
100-200
100-200
60
s
60
s
10
PUFF
10
PUFF
| title | di.xpt (a study reference domain for unique device identification) |
|---|
di.xpt (copied v1.0 of medical devices IG)
- This example is a copy of Example 1 from di.xpt in SDTMIG-MD v1.0 but with values for SPDEVID and DIVAL revised slightly.
- Should I remove the FDA UDI (row 5) unless CTP has or plans to establish UDI values?
Example 1
This shows records for two three devices where the sponsor felt that the type, manufacturer, model number, and serial number were necessary for unique identification. In addition, there was a post-marketing UDI identifier available for the first device.
- Rows 1-5 show the records for a device given a SPDEVID of PUFFMASTER3K
- Rows 5-8 show the records for a device given a SPDEVID of PUFFMASTER2023
- Rows 9-12 show
Row | STUDYID | DOMAIN | SPDEVID | DISEQ | DIPARMCD | DIPARM | DIVAL |
1 | 123 | DI | PUFFMASTER3K | 1 | DEVTYPE | Device Type | ENDS |
2 | 123 | DI | PUFFMASTER3K | 2 | MANUF | Manufacturer | Acme Machines |
3 | 123 | DI | PUFFMASTER3K | 3 | MODEL | Model Number | 45-JFI |
4 | 123 | DI | PUFFMASTER3K | 4 | SERIAL | Serial Number | 456789132-AXQ |
5 | 123 | DI | PUFFMASTER3K | 5 | FDAUDI | FDA Unique Device Identifier | 456789123xyz |
6 | 123 | DI | PUFFMASTER2023 | 1 | DEVTYPE | Device Type | SmokeMachine |
7 | 123 | DI | PUFFMASTER2023 | 2 | MANUF | Manufacturer | Acme Machines |
8 | 123 | DI | PUFFMASTER2023 | 3 | MODEL | Model Number | 62-PLC |
9 | 123 | DI | PUFFMASTER2023 | 4 | SERIAL | Serial Number | 215964564-NFS |
10 | 123 | DI | UsualCTCigarette | 1 | DEVTYPE | Device Type | Combustible Tobacco Cigarette |
11 | 123 | DI | UsualCTCigarette | 2 | MANUF | Manufacturer | Philip Morris International |
12 | 123 | DI | UsualCTCigarette | 3 | MODEL | Model Number | Marlboro Red |
13 | 123 | DI | UsualCTCigarette | 4 | SERIAL | Serial Number | 123456789 |
| title | ei.xpt Entity Identifiers (dm-like) |
|---|
- Entity IDs are unique within the assay (and a study can have many assays)
- Replicates vs. Run
- need planned (ei.xpt) separate from the actual (gt.xpt)
- If tests run at the same time, they are replicates and are assigned the same RUNID
- If tests are run at separate times, they are runs and the RUNIDs are distinct
- Different runs would be expected to have different ENID and GTDTC values. In ei.xpt, these are distinguished by different RUNID values.
- Entities that are replicates of each other would be expected to
- have different ENID values
- be in the same trial set (SETCD values)
- have the same RUNID values
RUNID
REPNUM (perm)
1
1
1
1
2
1
1
1
1
2
| title | gt.xpt (similar to LB) |
|---|
A1:
A2:
- Different runs would be expected to have different ENID and GTDTC values. In ei.xpt, these are distinguished by different RUNID values.
(cells evaluated)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|