...
| Dataset wrap |
|---|
|
| Rowcaps |
|---|
| Rows 1-23: | Show trial set parameters and values that comprise the test conditions for trial set A1. Set A1 is the data for the negative control (concentration 0) with short-term exposure and metabolic activation S9. The applicant has chosen to given a long name (SET) equal to "ST+S9_C0". Set A1 is associated with the first row in the sample report table for study 123. | | Rows 24-46: | Show trial set parameters and values that comprise the test conditions for trial set A2. Set A2 is the data for the short-term exposure with metabolic activation S9 at a concentration of 1250 ug/ml. The applicant has chosen to give the set a long name (SET) equal to "ST+S9_C1250". Set A2 is associated with the second row in the sample report table for study 123. |
|
| Dataset2 |
|---|
TSSEQTSGRPIDTSPARMCDTSPARMTSVALTSVALNFTS| SSTYP | Study Type | REPEAT DOSE TOXICITY | 2 | TS | 1 | SPECIES | Species | RAT | 3 | TS | 1 | STRAIN | Strain/Substrain | FISCHER 344 | 4 | TS | 1 | SPLRNAM | Test Subject Supplier | HARLAN | 5 | TS | 1 | SDESIGN | Study Design | CROSSOVER | 6 | TS | 1 | ROUTE | Route of Administration | ORAL | 7 | TS | 1 | GLPTYP | Good Laboratory Practice Type | FDA | 8 | TS | 1 | EXPSTDTC | Experimental Start Date | 2008-01-01 | 9 | TS | 1 | EXPENDTC | Experimental End Date | 2008-03-07 | 10 | TS | 1 | TRMSAC | Time to Terminal Sacrifice | P42D | 11 | TS | 1 | STSTDTC | Study Start Date | 2007-12-30 | 12 | TS | 1 | DOSDUR | Dosing Duration | P42D | 13 | TS | 1 | STITLE | Study Title | Example of a Crossover study in the Rat with 3 dose levels and 3 dosing periods | 14 | TS | 1 | SNDIGVER | SEND Implementation Guide Version | SEND IMPLEMENTATION GUIDE VERSION 3.2 | 15 | TS | 1 | SNDCTVER | SEND Controlled Terminology Version | SEND Terminology 2021-03-26 | 16 | TS | 1 | STCAT | Study Category | TOX | 17 | TS | 1 | SSPONSOR | Sponsor Organization | Example Sponsor Inc. | 18 | TS | 1 | SPREFID | Sponsor's Study Reference ID | NOT APPLICABLE | 19 | TS | 1 | 1 | TSTFNAM | Test Facility Name | Example Tox Lab Name | 20 | TS | 1 | 1 | TSTFLOC | Test Facility Location | 10 Somewhere Street, Montgomery, AL 10000 | 21 | TS | 1 | 1 | TFCNTRY | Test Facility Country | USA | 22 | TS | 1 | AGETXT | Age Text | 6-8 | 23 | TS | 1 | AGEU | Age Unit | WEEKS | 24 | TS | 1 | 1 | STDIR | Study Director | Dr. R. Smith | 25 | TS | 1 | TRT | Investigational Therapy or Treatment | Drug A | 26 | TS | 1 | 2 | TRTV | Treatment Vehicle | Saline | 27 | TS | 1 | 2 | TRTVDESC | Treatment Vehicle Structured Description | 100.46 %(w/v) ISOTONIC SODIUM CHLORIDE SOLUTION {UNII VR5Y7PDT5W} | 28 | TS | 1 | GLPFL | GLP Flag | Y | 29 | TS | 1 | TRTCAS | Primary Treatment CAS Registry Number | TEMPORARILY UNAVAILABLE | | | Expand |
|---|
|
| Expand |
|---|
|
| Row | STUDYID | ASSAYID | DOMAIN | ARMCD | ARM | TAETORD | ETCD | ELEMENT |
|---|
1 | TA | ARM01-Prod1 | Cigarette Smoke Condensate, 10 ug/mL | 1 | TREATMT | Treatment | 9 | 9 | SLS-110 | 1 | | Expand |
|---|
| title | te.xpt (trial elements) |
|---|
|
| Row | STUDYID | ASSAYID | DOMAIN | ETCD | ELEMENT |
|---|
TE | TREATMT | Treatment | | Expand |
|---|
|
| Expand |
|---|
|
Red font (CT is coming!, POSITIVE CONTROL, VEHICLE CONTROL, etc.)
ARMCD. TREATMENT (element). SETCD
ARM01 TREATMT. CS-01. (Cigarette Smoke 10 ug/mL)
ARM01
TA.XPT
TRIAL ARMS
| Row | STUDYID | ASSAYID | DOMAIN | ARMCD | ARM | TAETORD | ETCD | ELEMENT |
|---|
1 | TA | ARM01-Prod1 | Cigarette Smoke Condensate, 10 ug/mL | 1 | TREATMT | Treatment | 2 | TA | ARM02-Prod1 | Cigarette Smoke Condensate, 50 ug/mL | 1 | TREATMT | Treatment | 3 | TA | ARM03-Prod1 | Cigarette Smoke Condensate, 75 ug/mL | 1 | TREATMT | Treatment | 4 | TA | 4 | 1 | 5 | TA | 5 | 1 | 6 | TA | 6 | 1 | 7 | 7 | 1 | 8 | 8 | 1 | 9 | 9 | SLS-110 | 1 | 10 | ARM10 | SLS-200 | 1 | TE.XPT
TRIAL ELEMENTS
| Row | STUDYID | ASSAYID | DOMAIN | ETCD | ELEMENT |
|---|
TE | TREATMT | Treatment | tx.spt
TRIAL SET
example: Each plate has a separate product
Product, Smoking Regime (e.g. Traditional combustible, ENDS), Smoke Fraction, etc.
https://www.itis.gov/servlet/SingleRpt/SingleRpt?search_topic=TSN&search_value=969610#null
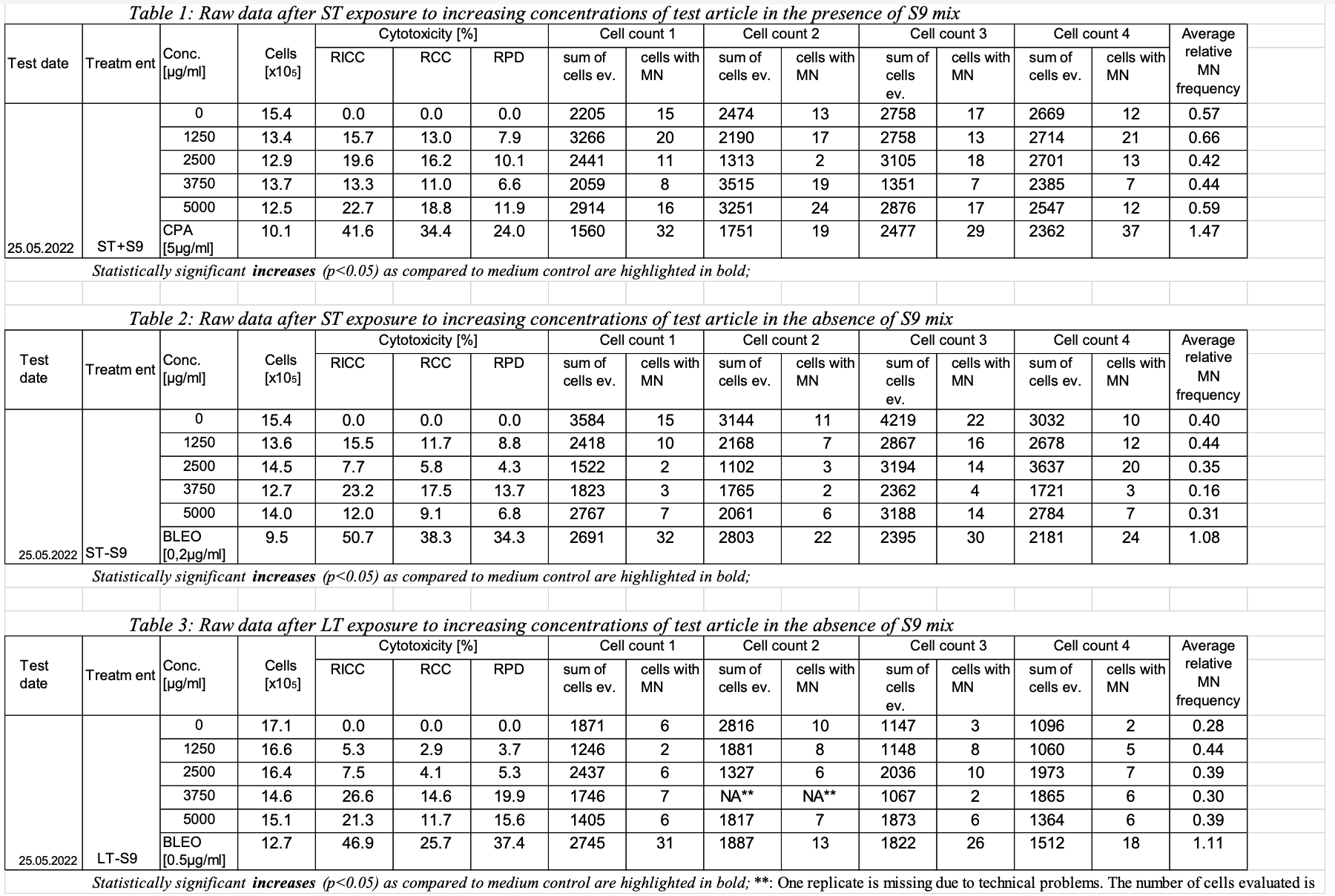
One study (Original sample of TK6 cells); (6 rows in GT)
Assay (MNT) (ran on 10 tobacco products)
3 assay/test conditions;
Treatment duration (2: ST, LT);
Presence of metabolic activation (2: presence, type)
Concentrations of product (6 of these: 0, x, y, z,..., +CNTL);
CytoTox % (3 different percentages from testing 1 portion of the original sample)
4 MN counts conducted (on 4 different portions of original sample?)
EID (5 of these, Count1 - Count4 and Cytotox): with many TX Parm codes
Cytotox: RICC, RIC, RPD, #cells
| Row | STUDYID | ASSAYID | DOMAIN | SETCD | SET | TXSEQ | TXPARMCD | TXPARM | TXVAL |
|---|
1 | 123 | 1 | TX | METACT | Metabolic Activation | +S9 | 2 | METACT | Metabolic Activation | -S9 | TRTDUR | Treatment Duration | Short Term | TRTDUR | Treatment Duration | Long Term (or 2 parm codes, # and unit) | PRDCONC | Concentration of product | 0 | PRDCONCU | Concentration Unit | ug/ml | Product | Product Name | Smoking Regime | Smoke Fraction | TX | CS-10 | Cigarette Smoke Condensate, 10 ug/mL | ARMCD | Arm Code | CS-10 | TX | CS-10 | Cigarette Smoke Condensate, 10 ug/mL | SPECIES | Species | Salmonella enterica | TX | CS-10 | Cigarette Smoke Condensate, 10 ug/mL | STRAIN | Strain/Substrain | Salmonella enterica enterica | TX | CS-10 | Cigarette Smoke Condensate, 10 ug/mL | PRODUCT | Product | product A | TX | CS-10 | Cigarette Smoke Condensate, 10 ug/mL | REGIME | Smoking Regime | Traditional combustible | TX | CS-20 | Cigarette Smoke Condensate, 50 ug/mL | ARMCD | Arm Code | CS-20 | TX | CS-20 | Cigarette Smoke Condensate, 50 ug/mL | PRODUCT | Product | product A | ... | TX | LSL-200 | sodium lauryl sulphate (SLS) positive control, 200 ug/mL | TRTDOS | Dose Level | 100 | TX | LSL-200 | sodium lauryl sulphate (SLS) positive control, 200 ug/mL | TCNTRL | Control Type | POSITIVE CONTROL | TX | LSL-200 | sodium lauryl sulphate (SLS) positive control, 200 ug/mL | CNTLAG | Control Agent | SODIUM LAURYL SULPHATE | TX | LSL-200 | sodium lauryl sulphate (SLS) positive control, 200 ug/mL | CNTLAGAMT | Control Agent amount (levels) | 110 | LSL-200 | sodium lauryl sulphate (SLS) positive control, 200 ug/mL | CNTLAGUNIT | Control Agent Unit | ug/mL | | Expand |
|---|
|
Row | STUDYID | DOMAIN | ENID (Entity ID) | RICC | RCC | RPD | Sum of cell ev. | Cells with MN | SMKFID (Smoke Fraction) | REPLCTID (Replicate Number) | PLATEID (Plate ID) | COLID (Column number) | ROWID (Row number) | SETCD (Set Code, TX) | RFSTDTC | RFENDTC | RFXSTDTC | RFXENDTC | RFCSTDTC | RFCENDTC | ARMCD | 1 | MA99999 | GT | 030001 | A | 1 | 1 | 1 | A | CS-10 | 2008-04-01T06:00 | | 2008-04-02T14:00 | 2008-04-02T14:04 | 2008-04-01T06:00 | 2008-04-01T06:00 | 3 | 2 | MA99999 | GT | 1 | 1-1 | 030001 | A | 1 | 1 | 1 | B | CS-10 | 2008-04-01T06:00 | 2008-04-21T14:00 | 2008-04-02T20:00 | 2008-04-02T20:04 | 2008-04-01T06:00 | 2008-04-01T06:00 | 3 | 2 | 3 | 4 |