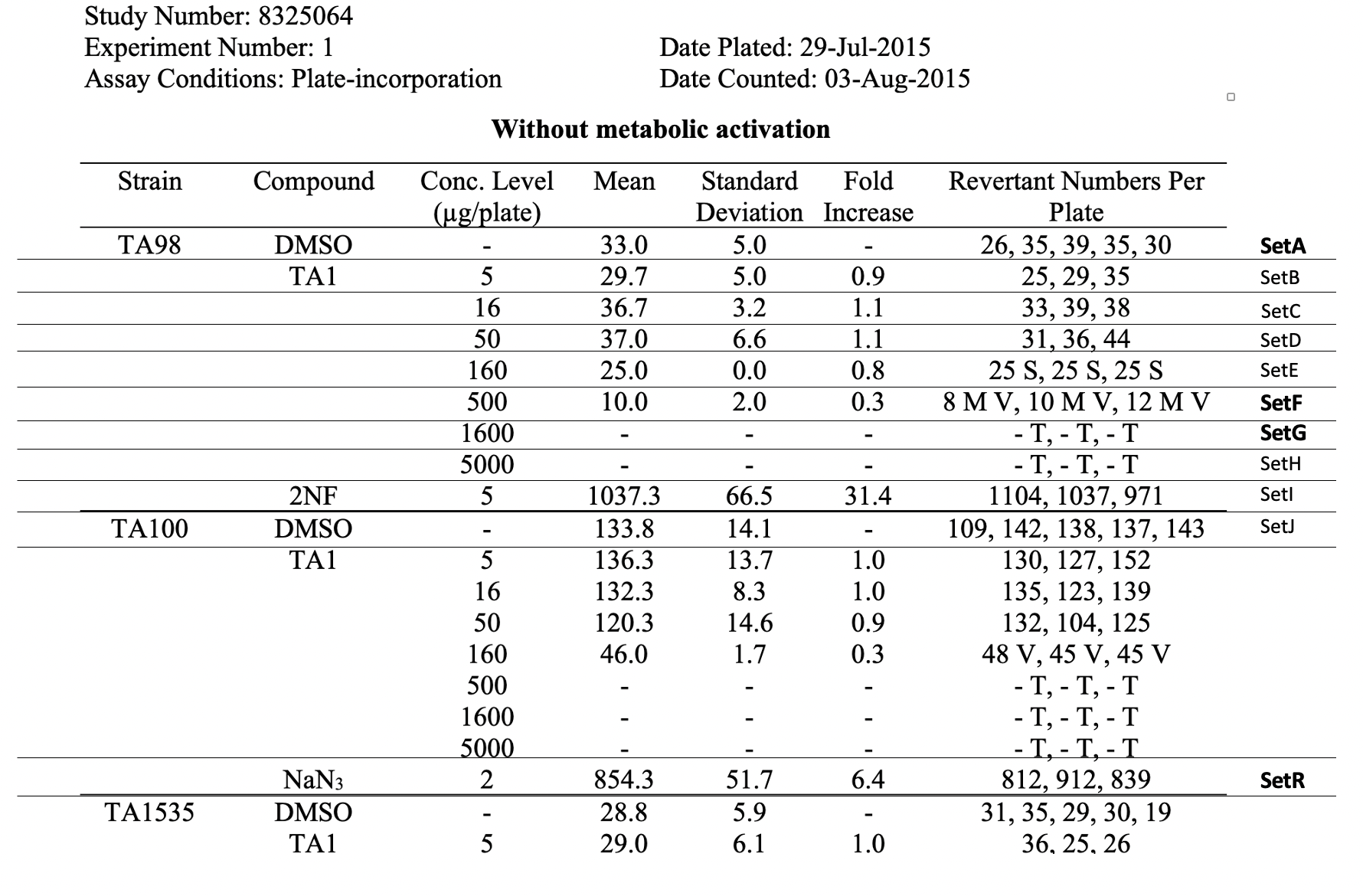
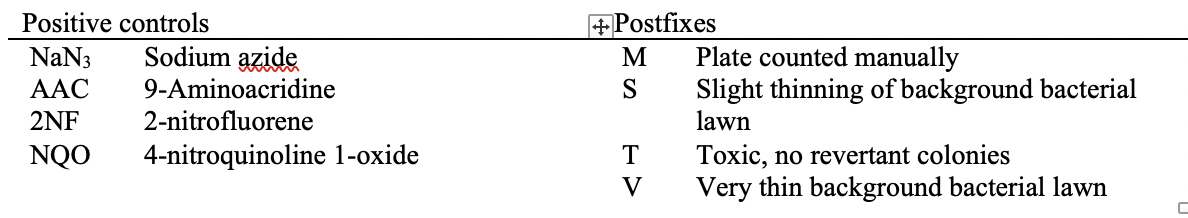
| SMKRGM | Smoking Regimen | NON-INTENSE REGIMEN | | 15 | 8325064 | TX | SetA | A-TA98-C0 | 15 | STRAIN | Strain/Substrain | TA98 | | 16 | 8325064 | TX | SetA | A-TA98-C0 | 16 | MTACTIND | Metabolic Activating Agent Name | NOT APPLICABLE | | 17 | 8325064 | TX | SetA | A-TA98-C0 | 17 | METACTFL | Presence of Metabolic Activation Flag | N | | 18 | 8325064 | TX | SetA | A-TA98-C0 | 18 | ITVNAM | Intervention Article Name | DMSO | | 19 | 8325064 | TX | SetA | A-TA98-C0 | 19 | ITVTYPE | Intervention Article Type | VEHICLE | | 20 | 8325064 | TX | SetA | A-TA98-C0 | 20 | ITVCONC | Intervention Article Concentration | 100 | | 21 | 8325064 | TX | SetA | A-TA98-C0 | 21 | ITVCONCU | Intervention Article Concentration Unit | % | | 22 | 8325064 | TX | SetA | A-TA98-C0 | 22 | TRTV | Treatment Vehicle | DMSO | | 23 | 8325064 | TX | SetF | F-TA98-C500 | 23 | SPECIES | Species | SALMONELLA TYPHIMURIUM | | 24 | 8325064 | TX | SetF | F-TA98-C500 | 24 | IVTDMIN | In vitro Treatment Duration Minimum | 71.5 | | 25 | 8325064 | TX | SetF | F-TA98-C500 | 25 | IVTDTRG | In vitro Treatment Duration Target | 72 | | 26 | 8325064 | TX | SetF | F-TA98-C500 | 26 | IVTDMAX | In vitro Treatment Duration Maximum | 72.5 | | 27 | 8325064 | TX | SetF | F-TA98-C500 | 27 | IVTDU | In vitro Treatment Duration Unit | HOURS | | 28 | 8325064 | TX | SetF | F-TA98-C500 | 28 | INCBTMP | Incubation Temperature | 37 | | 29 | 8325064 | TX | SetF | F-TA98-C500 | 29 | INCBTMPU | Incubation Temperature Unit | C | | 30 | 8325064 | TX | SetF | F-TA98-C500 | 30 | ATMRHP | Atmospheric Relative Humidity Percent | 50 | | 31 | 8325064 | TX | SetF | F-TA98-C500 | 31 | ATMCO2P | Atmospheric CO2 Percent | 5 | | 32 | 8325064 | TX | SetF | F-TA98-C500 | 32 | SPTOBID | Applicant-defined tobacco identifier | CIG01a | | 33 | 8325064 | TX | SetF | F-TA98-C500 | 33 | EXPTYP | | Submerged | | 34 | 8325064 | TX | SetF | F-TA98-C500 | 34 | SAMTYP | Sample Type | Total Particulate Matter in PBS | | 35 | 8325064 | TX | SetF | F-TA98-C500 | 35 | APDEVID | Applicant-defined device identifier | PUFFMASTER3K | | 36 | 8325064 | TX | SetF | F-TA98-C500 | 36 | SMKRGM | Smoking Regimen | NON-INTENSE REGIMEN | | 37 | 8325064 | TX | SetF | F-TA98-C500 | 37 |
|
|
| Row | STUDYID | ASSAYID | DOMAIN | SETCD | SET | TXSEQ | TXPARMCD | TXPARM | TXVAL |
|---|
| 1 | 8325064 | Ames | TX | SetA | A-TA98-C0 | 1 | SPECIES | Species | Salmonella typhimurium |
2 | 8325064 | Ames | TX | SetA | A-TA98-C0 | 2 | STRAIN | Strain/Substrain | TA98 |
|
3AmesTXASetA | C03METACT Activation | Activating Agent Name | NOT APPLICABLE | | 39 |
|
4AmesSetA AC04Y/N presence of metabolic activation| Presence of Metabolic Activation Flag | N |
|
5Ames | SetAAC05TRTDMIN | Treatment Duration Minimum | 71.5 | 6| ITVNAM | Intervention Article Name | TA1 | | 41 | 8325064 |
|
Ames | SetAAC06TRTDTRG | Treatment Duration Target | 72 | 7| ITVTYPE | Intervention Article Type | PRODUCT | | 42 | 8325064 |
|
Ames | SetAAC07TRTDMAX | Treatment Duration Maximum | 72.5 | | ITVCONC | Intervention Article Concentration | 500 | | 43 | 8325064 |
|
8 | 8325064 | AmesSetAAC08TRTDU | | ITVCONCU | Intervention Article Concentration |
|
Treatment Duration HOURS9AmesSetAAC09INCBTMPIncubation Temperature3710AmesSetAAC010INCBTMPUIncubation Temperature Unit | C | 11| Species | SALMONELLA TYPHIMURIUM | | 46 | 8325064 |
|
Ames | SetAAC011HUMID | Atmospheric Relative Humidity Percent | 50 | 12| IVTDMIN | In vitro Treatment Duration Minimum | 71.5 | | 47 | 8325064 |
|
Ames | SetAAC012ATMCO2 | Atmospheric CO2 Percent | 5 | 13| IVTDTRG | In vitro Treatment Duration Target | 72 | | 48 | 8325064 |
|
Ames | SetAAC013SPTOBID | Sponsor defined tobacco identifier | CIG01a | 14| IVTDMAX | In vitro Treatment Duration Maximum | 72.5 | | 49 | 8325064 |
|
Ames | SetAAC014EXPTYP | Exposure Type (See TIG NC workstream minutes 30-Jan here: Nonclinical) | Submerged | | IVTDU | In vitro Treatment Duration Unit | HOURS | | 50 |
|
15Ames | SetAAC015SAMTYP Sample Type | Total Particulate Matter in PBS | | Incubation Temperature | 37 | | 51 |
|
16Ames | SetAAC016INTRVN | name of the intervention article | TA1 | 17 | 8325064 | | INCBTMPU | Incubation Temperature Unit | C | | 52 | 8325064 |
|
AmesSetAAC017ITVTYPE | type of intervention article | Vehicle Control | 18 | 8325064 | | ATMRHP | Atmospheric Relative Humidity Percent | 50 | | 53 | 8325064 |
|
AmesSetAAC018ITVCONC | Concentration of intervention article | 0 | | ATMCO2P | Atmospheric CO2 Percent | 5 | | 54 |
|
19Ames | SetAAC019ITVCONCU | Concentration Unit | ug/plate | | SPTOBID | Applicant-defined tobacco identifier | CIG01a | | 55 |
|
20Ames | SetAAC020TRTVTreatment VehicleDMSO21Ames | SetAAC021SPDEVID | Sponsor defined device identifier | PUFFMASTER3K | | SAMTYP | Sample Type | Total Particulate Matter in PBS | | 57 | 8325064 |
|
22 | 8325064 | AmesSetAAC022DUREFID | Smoke Regimen | Medium Intensity Regimen | | APDEVID | Applicant-defined device identifier | PUFFMASTER3K | | 58 |
|
23Ames | SetFFC5001SPECIES | Species | | Smoking Regimen | NON-INTENSE REGIMEN | | 59 |
|
24Ames | SetFFC5002| 59 | STRAIN | Strain/Substrain | TA98 |
|
25Ames | SetFFC5003METACT Activation | Activating Agent Name | NOT APPLICABLE | | 61 |
|
26Ames | SetFFC5004Y/N presence of metabolic activation| Presence of Metabolic Activation Flag | N |
|
27Ames | SetFFC500TRTDRTOL | Treatment Duration Tolerance | 28 | 8325064 | | C1600 | 62 | ITVNAM | Intervention Article Name | TA1 | | 63 | 8325064 |
|
AmesSetFFC500TRTDURU | Treatment Duration Unit (this is for both TRTDURT, TRTDURTOL)| C1600 | 63 | ITVTYPE | Intervention Article Type | Product | | 64 | 8325064 |
|
Ames | SetFFC500INTRVN | name of the intervention article (Tobacco ProdA, Bleomycin or Cyclophosphamid A) | TA1 | 8325064 | | ITVCONC | Intervention Article Concentration | 1600 | | 65 | 8325064 |
|
AmesSetFFC500ITVTYPE | type of intervention article choices of values: product; negative control; positive control | Product | 8325064 | Ames | TX | SetF | F-TA98-C500 | ITVCONC | Concentration of intervention article | 500 | 8325064 | Ames | TX | SetF | F-TA98-C500 | ITVCONCU | Concentration Unit | ug/plate | 8325064 | Ames | TX | SetF | F-TA98-C500 | STRAIN | Strain/Substrain | TA98 | 8325064 | Ames | TX | SetF | F-TA98-C500 | REGIME | Smoking Regime | ISO Regime | ... | 8325064 | Ames | TX | SetG (row 7) do G not I!!! | METACT | Metabolic Activation (should there be two parms? Presence, type)? | None | 8325064 | Ames | TX | SetG | TRTDRTRG | Treatment Duration target. (how do we show 3-6 hour range? start/end, target and tolerance?, one text field not-analyzable) | 8325064 | Ames | TX | SetG | TRTDRTOL | Treatment Duration Tolerance | 8325064 | Ames | TX | SetG | TRTDURU | Treatment Duration Unit (this is for both TRTDURT, TRTDURTOL) | 8325064 | Ames | TX | SetG | INTRVN | name of the intervention article (Tobacco ProdA, Bleomycin or Cyclophosphamid A) | 2-Nitrofluorine | 8325064 | Ames | TX | SetG | ITVTYPE | type of intervention article choices of values: product; negative control; positive control | Positive Control | 8325064 | Ames | TX | SetG | ITVCONC | Concentration of intervention article | 5 | 8325064 | Ames | TX | SetG | ITVCONCU | Concentration Unit | ug/plate | 8325064 | Ames | TX | SetG | REGIME | Smoking Regime | ISO | ... | SetG | 8325064 | Ames | TX | SetR (Row 18) | METACT | Metabolic Activation (should there be two parms? Presence, type)? | None | 8325064 | Ames | TX | SetR | TRTDRTRG | Treatment Duration target. (how do we show 3-6 hour range? start/end, target and tolerance?, one text field not-analyzable) | 8325064 | Ames | TX | SetR | TRTDRTOL | Treatment Duration Tolerance | 8325064 | Ames | TX | SetR | TRTDURU | Treatment Duration Unit (this is for both TRTDURT, TRTDURTOL) | 8325064 | Ames | TX | SetR | INTRVN | name of the intervention article (Tobacco ProdA, Bleomycin or Cyclophosphamid A) | 4-Nitroquinoline-1-oxide | 8325064 | Ames | TX | SetR | ITVTYPE | type of intervention article choices of values: product; negative control; positive control | Positive Control | 8325064 | Ames | TX | SetR | ITVCONC | Concentration of intervention article | 2 | 8325064 | Ames | TX | SetR | ITVCONCU | Concentration Unit | ug/plate | 8325064 | Ames | TX | SetR | STRAIN | Strain/Substrain | TA98 | 8325064 | Ames | TX | SetR | REGIME | Smoking Regime | ISO |